 Here at Outdoors with Othmar Vohringer and Whitetail Deer Passion we recognizes the current threat that chronic wasting disease (CWD) poses for the deer, elk and perhaps other wildlife plus to the heritage of hunting big game in general that could occur if this illness should spread further. In several regions of North America this threat has become already a sad reality. This is why we here at Outdoors with Othmar Vohringer and Whitetail Deer Passion have compiled an informative resource of information so that the visitors to our website can learn more about this threat and what they can do to help in the prevention of the spread of CWD.
Here at Outdoors with Othmar Vohringer and Whitetail Deer Passion we recognizes the current threat that chronic wasting disease (CWD) poses for the deer, elk and perhaps other wildlife plus to the heritage of hunting big game in general that could occur if this illness should spread further. In several regions of North America this threat has become already a sad reality. This is why we here at Outdoors with Othmar Vohringer and Whitetail Deer Passion have compiled an informative resource of information so that the visitors to our website can learn more about this threat and what they can do to help in the prevention of the spread of CWD.As hunters and conservationists we at Outdoors with Othmar Vohringer and Whitetail Deer Passion feel it is our duty to our fellow outdoor and hunting enthusiasts to provide the information available without the usual hype, rumours and hearsay. Instead we provide the facts, as they are known to us from reputable sources in an effort that you can take the necessary precautions and to learn what various state and province agencies in the USA and Canada are doing to combat this problem. For most hunters, especially those who hunt in areas where CWD has not been found do not be concerned about CWD and still can enjoy the upcoming hunting season. But we at Othmar Vohringer Outdoors would like to encourage all hunters who care about wildlife and conservation as much as we do, to get as much information and education about this illness as possible, to take all the necessary precautions and help their local wildlife management agencies in any way they see fit in preventing the spread of CWD in an effort to secure our priceless natural renewable resources for generations to come.. Thank you for being a true hunter and conservationist, we count on you and so do our children and future generation hunters.
Bellow we provide you with the answers to most CWD questions.
**************
 What is CWD?
What is CWD?CWD is a neurological (brain and nervous system) disease found in deer and elk in certain geographical locations in North America. The disease belongs to a family of diseases known as transmissible spongiform encephalopathises (TSE) or prion diseases.
This disease attacks the brain of the infected deer or elk and produces small lesions that result in the death of the animal. While CWD is similar to mad cow disease (BSE) in cattle and as scrapie in sheep, there is no known relationship between CWD and any other TSE of animals and, or people
.
How is it spread?
It’s not known exactly how CWD is spread. It is believed that the agent responsible for the disease may be spread both directly (animal to animal) and indirectly (soil or other surface to animal). It is thought that the most common mode of transmission from an infected animal is via saliva and feces.
Where has it been found?
CWD is known to infect wild deer and elk in north-eastern Colorado and southern Wyoming, and wild deer in western Colorado, western Nebraska, south-western South Dakota, south-central New Mexico and west-central Saskatchewan. It has been diagnosed in game ranches in Colorado, Nebraska, South Dakota, Montana, Oklahoma, Kansas, Alberta and Saskatchewan.
Is it dangerous to humans?
There is currently no convincing evidence that the agent of CWD affects humans. However, public health officials recommend that human exposure to CWD agent be avoided as they continue to research the disease.
What precautions should hunters take?
Health officials advise hunters not to consume meat from animals known to be infected with the disease. Boning out meat is recommended. In addition, they suggest that hunters take normal simple hygienic precautions when field dressing carcasses.
How can you tell if a deer has CWD?
Infected animals may not show any symptoms of the disease. In some stages of the disease, however, infected animals begin to lose bodily functions and display abnormal behaviour such as staggering, disorientation or standing with very pore posture. Animals may have an exaggerated wide posture, or may carry the head and ears lowered. Infected animals become very emaciated (thus wasting disease) and will appear in very poor body condition. Infected animals will also often stand near water. Drooling or excessive salivation may be apparent. Note that these symptoms may also be characteristic of diseases other than CWD.
What should I do if I see a deer with CWD?
Unless your state or province wildlife agency or other appropriate authority has issued other instructions and/or regulations, you should accurately document the location of the animal and immediately contact the nearest officer or employee of the State or Province Wildlife Agency. Do not attempt to contact, disturb, kill, or remove the animal.
Can I have a deer tested?
In general, the answer is no if you live in an area where CWD has not been documented. Testing is currently only available in areas where CWD has been detected in wild deer. Much effort is being devoted to increasing the laboratories that are certified to do the testing.
Is the meat safe to eat?
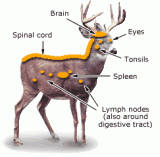
While the agent that produces chronic wasting disease in deer and elk has not been positively identifies, there is strong evidence to suggest that abnormally shape proteins, called prions, are responsible. Research completed to date indicate that the prions accumulate in certain parts of infected animals-the brain, eyes, spinal cord, lymph nodes, tonsils and spleen. Based on these findings, hunters are recommended to not eat meat from animals known to be infected with CWD. Hunters in CWD areas are also advice to bone out their meat and to not consume those parts where prions likely accumulate.
What's being done to combat CWD?
Efforts to address CWD are accelerating rapidly. In facilities with captive animals known to have or to have been exposed to CWD, management is concentrating on quarantining or killing of every animal and burning of all carcasses. In some cases around captive populations, double fencing is recommended to prevent direct contact between captive and wild animals.
In wild populations, the management option recommended is to reduce the density of animals in the infected area to slow the transmission of the disease. This is being done by selective culling of animals suspected to have been directly exposed to this disease. In Colorado, Nebraska and Wisconsin large numbers of animals are being killed to reduce density of animals and thus slow the transmission of the disease.
There is still a large need for research on the disease as many questions go unanswered. There s also a need for increased funding to support additional laboratories for testing animals for the disease. Just about every U.S. State and Canadian Province wildlife agency is now planning an increased effort at surveillance to detect if CWD is present.
Many state and province agencies have banned the importation of deer and elk into their state or province.
Some U.S. States and Canadian Provinces have also halted intra-state/province movement of deer and elk has banned supplemental feeding programs. Colorado has implemented regulations that allow only boned meat, quarters (without spinal column or head) or processed meat from deer or elk to be transported out of certain areas with CWD. Clean skull plates with the antlers attached can also be removed from an infected area. Check with a state/province wildlife agency in the state/province where you hunt to determine if they have enacted similar restrictions.
For more up-to-date information please visit:
The CWD.Org. Website
Chronic Wasting Disease is a joint project of the Boone and Crockett Club, the Mule Deer Foundation and the Rocky Mountain Elk Foundation. These non-profit wildlife conservation organizations formed the Chronic Wasting Disease Alliance in January 2002 to address CWD. Other organizations have since joined the Alliance.
Updates about CWD on this Blog:
West Virginia CWD
Infectious Prions Found in Deer Meat

2 comments:
0C7.04
North American Cervids Harbor Two Distinct CWD Strains
Authors
Angers, R. Seward, T, Napier, D., Browning, S., Miller, M., Balachandran A., McKenzie, D., Hoover, E., Telling, G. 'University of Kentucky; Colorado Division of Wildlife, Canadian Food Inspection Agency; University Of Wisconsin; Colorado State University.
Content
Despite the increasing geographic distribution and host range of CWD, little is known about the prion strain(s) responsible for distinct outbreaks of the disease. To address this we inoculated CWD-susceptible Tg(CerPrP)1536+/· mice with 29 individual prion samples from various geographic locations in North America. Upon serial passage, intrastudy incubation periods consistently diverged and clustered into two main groups with means around 210 and 290 days, with corresponding differences in neuropathology. Prion strain designations were utilized to distinguish between the two groups: Type I CWD mice succumbed to disease in the 200 day range and displayed a symmetrical pattern of vacuolation and PrPSc deposition, whereas Type II CWD mice succumbed to disease near 300 days and displayed a strikingly different pattern characterized by large local accumulations of florid plaques distributed asymmetrically. Type II CWD bears a striking resemblance to unstable parental scrapie strains such as 87A which give rise to stable, short incubation period strains such as ME7 under certain passage conditions. In agreement, the only groups of CWD-inoculated mice with unwavering incubation periods were those with Type I CWD. Additionally, following endpoint titration of a CWD sample, Type I CWD could be recovered only at the lowest dilution tested (10-1), whereas Type II CWD was detected in mice inoculated with all dilutions resulting in disease. Although strain properties are believed to be encoded in the tertiary structure of the infectious prion protein, we found no biochemical differences between Type I and Type II CWD. Our data confirm the co·existence of two distinct prion strains in CWD-infected cervids and suggest that Type II CWD is the parent strain of Type I CWD.
see page 29, and see other CWD studies ;
http://www.neuroprion.org/resources/pdf_docs/conferences/prion2008/abstract-book-prion2008.pdf
Sunday, November 23, 2008
PRION October 8th - 10th 2008 Book of Abstracts
http://bse-atypical.blogspot.com/2008/11/prion-october-8th-10th-2008-book-of.html
Biochim Biophys Acta. Author manuscript; available in PMC 2008 December 9. Published in final edited form as: Biochim Biophys Acta. 2007 June; 1772(6): 681-691. Published online 2006 December 15. doi: 10.1016/j.bbadis.2006.12.006. PMCID: PMC2597801 NIHMSID: NIHMS25810
Copyright notice and Disclaimer
The prion strain phenomenon: Molecular basis and unprecedented features
Rodrigo Morales,1,2 Karim Abid,1 and Claudio Soto1# 1 Protein Misfolding Disorders Laboratory, George and Cynthia Mitchell Center for Neurodegenerative diseases, Departments of Neurology, Neuroscience & Cell Biology and Biochemistry & Molecular Biology, University of Texas Medical Branch, 301 University Boulevard, Galveston, Texas, 77555-0646, USA 2 Facultad de Ciencias, Universidad de Chile, Santiago, Chile #To whom correspondence should be addressed at Email: clsoto@utmb.edu The publisher's final edited version of this article is available at Biochim Biophys Acta. See other articles in PMC that cite the published article.
Abstract
Prions are unconventional infectious agents responsible for transmissible spongiform encephalopathies. Compelling evidences indicate that prions are composed exclusively by a misfolded form of the prion protein (PrPSc) that replicates in the absence of nucleic acids. One of the most challenging problems for the prion hypothesis is the existence of different strains of the infectious agent. Prion strains have been characterized in most of the species. Biochemical characteristics of PrPSc used to identify each strain include glycosylation profile, electrophoretic mobility, protease resistance, and sedimentation. In vivo, prion strains can be differentiated by the clinical signs, incubation period after inoculation and the vacuolation lesion profiles in the brain of affected animals. Sources of prion strain diversity are the inherent conformational flexibility of the prion protein, the presence of PrP polymorphisms and inter-species transmissibility. The existence of the strain phenomenon is not only a scientific challenge, but it also represents a serious risk for public health. The dynamic nature and inter-relations between strains and the potential for the generation of a very large number of new prion strains is the perfect recipe for the emergence of extremely dangerous new infectious agents.
snip...
BSE has not only been transmitted to humans. The extensive use of cow-derived material for feeding other animals led to the generation of new diseases in exotic felines such as tiger and cheetah, non human primates, and domestic cats ?,57-60]. As it was mentioned before, the transmission of BSE into these different species could create many new prion strains, each one of them with particular biological and biochemical characteristics and thus a potentially new hazard for human health. Successful transmission of BSE in pigs has been described ?,62] and also in transgenic mice expressing pig PrP (PoPrP) ?]. Porcine derivates are widely consumed and the hypothetic case of "mad pigs" could increase the events of zoonotic transmission of prions to humans. Fortunately, transmission of BSE to pigs is possible only in very drastic conditions, not likely to be occurring naturally ?,63]. More frightening is perhaps the possibility that BSE has been passed into sheep and goats. Studies have already shown that this transmission is possible and actually relatively easy and worrisomely produces a disease clinically similar to scrapie ?]. The cattle origin of this new scrapie makes possible that the new strain may be transmissible to humans. Transmission experiments of BSE infected sheep brain homogenate into human transgenic animal models are currently ongoing in several laboratories. It is very important to note that all materials generated by transmission of BSE in experimental and natural cases show similar biochemical behavior compared to the original inoculum ?], suggesting that all these new generated infectious agents could potentially be hazardous for humans. The origin of BSE is still a mystery. Abundant evidence supports the hypothesis that BSE was produced by cattle feeding with scrapie derivated material ?,67], indicating that bovine PrPSc might be a "conformational intermediary" between ovine PrPSc and human PrPC.
There is currently no mean to predict which will be the conformation of a newly generated strain and how this new PrPSc conformation could affect other species. One interesting new prion disease is CWD, a disease affecting farm and wild species of cervids ?,69]. The origin of CWD and its potential to transmit to humans are currently unknown. This is worrisome, considering that CWD has became endemic in some parts of USA and the number of cases continues to increase ?]. It is presumed that a large number of hunters in the US have been in contact or consumed CWD-infected meat ?]. CWD transmissibility studies have been performed in many species in order to predict how this disease could be spread by consumption of CWD meat ?-73]. In these studies, a special attention has been done to scavenging animals ?], which are presumed to be exposed to high concentration of cervid prions, resulting in the putative generation of many new forms of TSEs. Fortunately negative results were obtained in one experiment done in raccoons infected with CWD ?]. Transmission of CWD to humans cannot be ruled out at present and a similar infective episode to BSE involving CWD could result in catastrophic events, spreading the disease in a very dangerous way through the human population. No clinical evidence linking CWD exposed humans and CJD patients have been found ?], but experimental inoculation of CWD prions into squirrel monkeys propagated the disease ?]. It is important to mention that the species barrier between humans and cervids appears to be greater than with cattle, as judged by experiments with transgenic mice models ?]. Finally, it is important to be aware about CWD transmissibility to other species in which a "conformational intermediary" could be formed, facilitating human infection.
SNIP...
VI. Unique features of prion strains
The biological and infectious characteristics of prions are dramatically different to the conventional infectious agents. These differences are manifested in the prion strains phenomenon in unique and unprecedented features, such as for example strain adaptation and memory, the coexistence and competition of prion strains, among others. In this section, some of these interesting phenomena will be briefly described.
Adaptation of Prion strains
Interspecies transmission of prions could result in the emergence of more than one variety of infectious material. All new collected infectious agents could present particular strain characteristics. That is the case of DY and HY prion strains generation ?,16]. When interspecies transmission of prions occurs, serial passages in the new host are needed in order to stabilize the characteristics of new generated infectious material. In the case of TME transmission in hamsters, at least four serial passages in the new species were required for stabilization ?]. The first passage was characterized by long incubation periods and a dominance of a 19 KDa fragment when newly obtained PrPSc was analyzed after PK digestion. In the three first passages, clinical symptoms were not characteristic of the hamster-adapted HY or DY TME strains. This phenotype was attributed to the combination effects of both strains replicating simultaneously. Thereafter, each of the strains was stabilized in some of the animals and once they are adapted and stabilized, they can be serially propagated in vivo and the characteristics are maintained. It is accepted that both strains present differential conversion kinetics in vitro, with DY being the slowest and HY the fastest ??]. For this reason, in order to select efficiently this prion strain, limit dilutions must be performed ?]. In that way, the most abundant and less convertible DY is favored against the less abundant but fastest HY strain.
Co-existence of prion strains
Related to the above, it has been shown that two or more prion strains can co-exist in natural cases of TSE. Co-existence of prion strains has been found in sporadic cases of CJD ??, 125]. Analyses of several sCJD tissue showed that different biochemical profiles of PrPSc could be found in different brain areas from the same patient ??]. Co-existence of prion strains was mainly observed in patient heterozygous for codon 129 ??]. As many as 50% of these patients present different types of PrPSc in their brains, whereas 9% of MM patients were positive for co-existence of strains. On the other hand, more than one PrPSc type was not observed in VV patients ??].
The biochemical and structural properties of the protein seem to be the major cause of this differential distribution. This observation may explain why sCJD is so heterogeneous in terms of clinical manifestation ?,126,127]. In a recent publication by Bishop et al. ??], vCJD infected transgenic mice expressing human PrPC, present changes in their PrPSc and vacuolation patterns in the brain according to their polymorphic classification for codon 129.
Competition of prion strains
In particular experimental conditions, some prion strains can extend their specific incubation period when co-infected with another strain. Long incubation period prions increase the incubation period of "faster" prions. This phenomenon of "competition of prion strains" has been observed in mice and hamster. In mice, competition between 22A and 22C strains was reported in 1975 by Dickinson et al. ??]. In this study, RIII mice (homozygous for sincs7 allele) were used. 22A and 22C showed long and short incubation period (550 and 230 days), respectively. When 22C strain was intraperitoneally inoculated 100, 200 and 300 days after intraperitoneal administration of the 22A agent, all three experimental groups resulted in Morales et al. Page 8 Biochim Biophys Acta. Author manuscript; available in PMC 2008 December 9. NIH-PA Author Manuscript NIH-PA Author Manuscript NIH-PA Author Manuscript incubation periods and lesion patterns matching 22A prions, suggesting that 22C prions were degraded or excreted, in animals previously infected by 22A. Similar results were obtained by Kimberlin and Walker in 1985 ??] using a different strain of sincs7 mice. These authors treated mice using 22A and 22C prion strain. Before inoculation, 22A was treated with different chemical and physical agents in order to see if the "competitor" or "blocking" characteristics of 22A were maintained. From all treatments, 12M urea was shown to almost abolish the blocking properties of 22A agent. This information suggests that infective properties of long incubation period agent are strictly necessary in order to increase the incubation period of faster prions.
In hamster, similar observations were reported using DY and HY ??]. DY prion strain was inoculated 30 and 60 days prior intraperitoneal inoculation of HY at three different doses. When incubation periods of HY inoculated control group were compared with the animals inoculated at 60 days with DY, significant differences in the incubation periods were found, especially when HY prions were administrated in a higher dose ??]. On the other hand no differences were observed in the case of intranerve inoculation, revealing that competition phenomenon occurs only when peripheral inoculation is performed. These results are surprising considering the fact that DY was reported not to be infectious when intraperitoneally inoculated in hamsters ??]. This data suggest that replication of DY is occurring in peripheral tissues but is not able to reach the central nervous system.
In general, the principal variables that need to be observed for a successful competition are the route of infection, the interval between injections and the particular strains and doses of agent used. Prolongation of incubation periods in TSE are therapeutically beneficial and several strategies are under development to reach this aim, including antibodies, beta-sheet breakers, and other chemical agents ??-133]. The experimental evidence described above suggests that prions could be potentially useful for this purpose. In order to prevent spread of prion disease in cattle or humans, prion strains with incubation periods longer than species' lifespan could be used to slowdown the replication of BSE or vCJD prions.
VII. Concluding Remarks
The existence of different strains of an infectious agent composed exclusively of a protein has been one of the most puzzling issues in the prion field. If is already difficult to understand how a protein can adopt two stable and different folded structures and that one of them can transform the other one into itself, it is unthinkable that the misfolded form can in turn adopt multiple conformations with distinct properties. Yet, compelling scientific evidence support the idea that PrP can adopt numerous folding patterns that can faithfully replicate and produce different diseases. The existence of the strain phenomenon is not only a scientific challenge, but it also represents a serious risk for public health. The dynamic nature and inter-relations between strains and the potential for the generation of many new prion strains depending on the polymorphisms and the crossing of species barrier is the perfect recipe for the emergence of extremely dangerous new infectious agents. Although, substantial progress has been made in understanding the prion strains phenomenon, there are many open questions that need urgent answers, including: what are the structural basis of prion strains?; how are the phenomena of strain adaptation and memory enciphered in the conformation of the prion agent?; to what species can a given prion strain be transmissible?; what other cellular factors control the origin and properties of prion strains?. ...SNIP...END
http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2597801&rendertype=abstract
http://www.pubmedcentral.nih.gov/picrender.fcgi?artid=2597801&blobtype=pdf
Friday, December 12, 2008
The prion strain phenomenon: Molecular basis and unprecedented features
http://bse-atypical.blogspot.com/2008/12/prion-strain-phenomenon-molecular-basis.html
Sunday, November 30, 2008 Commentary: Crimes hurt essence of hunting
http://chronic-wasting-disease.blogspot.com/2008/11/commentary-crimes-hurt-essence-of.html
TSS
Nice blog you got here. I'd like to read more concerning this theme. Thanx for sharing this information.
Sexy Lady
Busty London escorts
Post a Comment